
CIÊNCIAS FLORESTAIS
Physiographic and floristic gradients across topography in transitional seasonally dry evergreen forests of southeast Pará, Brazil
Gradientes Fisiográficos e Florísticos ao Longo do Relevo em Florestas Perenifólias Sazonalmente Secas de Transição no Sudeste do Pará, Brasil
James GROGAN1,2,*, Jurandir GALVÃO3
1Yale University School of Forestry & Environmental Studies, 360 Prospect Street, New Haven, CT 06511, USA;
2Instituto do Homem e Meio Ambiente da Amazônia (IMAZON), Caixa Postal 5101, Belém, Pará 66.613-397, Brasil;
3Mahogany Project Manager, Tauari/Capanema, Pará, Brasil; *Corresponding author: 44 Cave Hill Road, Leverett, MA 01054 USA; Tel: +1-413-548-8180; E-mail: jgrogan@imazon.org.br, jgrogan@crocker.com
ABSTRACT
Seasonally dry evergreen forests in southeast Pará, Brazil are transitional between taller closed forests of the interior Amazon Basin and woodland savannas (cerrados) of Brazil's south-central plains. We describe abiotic and biotic gradients in this region near the frontier town of Redenção where forest structure and composition grade subtly across barely undulating topography. Annual precipitation averaged 1859 mm between 1995-2001, with nearly zero rainfall during the dry season months of June August. Annual vertical migrations of deep-soil water caused by seasonal rainfall underlie edaphic and floristic differences between high- and low-ground terrain. Low-ground soils are hydromorphic, shaped by perching water tables during the wet season, pale gray, brown, or white in color, with coarse texture, low moisture retention during the dry season, and relatively high macro-nutrient status in the surface horizons. Forest canopies on low ground are highly irregular, especially along seasonal streams, while overstory community composition differs demonstrably from that on high ground. High-ground soils are dystrophic, well-drained through the wet season, brown or red-yellow in color, with finer texture, higher moisture retention, and low macro-nutrient status in the surface horizons compared to low-ground soils. Forest canopies are, on average, taller, more regular, and more closed on high ground. Low-ground areas can be envisioned as energy and nutrient sinks, where, because of hydrologic cycles, canopy disturbance likely occurs more frequently than at high-ground positions if not necessarily at larger scales.
KEY WORDS: Babaçu, big-leaf mahogany, disturbance, soil nutrients, water table
RESUMO
As florestas perenifólias sazonalmente secas no sudeste do Pará, Brasil, são áreas de transição entre as florestas fechadas mais altas do interior da Bacia Amazônica e o cerrado das planícies da região Sul-Central do Brasil. Descrevemos os gradientes abióticos e bióticos nessa região próxima da cidade de Redenção, onde a estrutura e a composição da floresta muda gradual e sutilmente ao longo da topografia levemente ondulada. Entre 1995 e 2001, o índice pluviométrico anual nessa região era de, em média, 1.859 mm, com aproximadamente zero de precipitação durante junho e agosto; os meses da estação seca. As migrações verticais anuais das águas profundas do solo, causadas pelas chuvas sazonais, são responsáveis pelas diferenças edáficas e florísticas entre os terrenos baixos e altos. Os solos dos terrenos baixos são hidromórficos formados pela água que se eleva do lençol freático durante a estação chuvosa de cor cinza pálido, marrom ou branca, textura grossa, com baixa retenção de umidade durante a estação seca e concentração relativamente alta de macro-nutrientes nos horizontes superficiais. O dossel florestal nos terrenos baixos é bastante irregular, especialmente ao longo dos igarapés sazonais, e a composição da comunidade do dossel difere visivelmente daquela dos terrenos altos. Os solos dos terrenos altos são distróficos, bem drenados durante a estação de chuvas, de cor marrom ou vermelho-amarela, textura fina, e possuem maior retenção de umidade e menor concentração de macro-nutrientes nos horizontes superficiais se comparados aos solos dos terrenos baixos. Nos terrenos altos, os dosséis florestais são, em média, mais altos, mais regulares e mais fechados. As áreas de terrenos baixos podem ser consideradas sumidouros de energia e nutrientes, nas quais, em virtude do ciclo hidrológico, as perturbações no dossel são prováveis de ocorrer mais freqüentemente do que nos terrenos mais altos, mas não necessariamente em escalas maiores.
PALAVRAS-CHAVE: babaçu, mogno, perturbação, nutrientes no solo, lençol freático
INTRODUCTION
The southeastern limits of closed Amazon forest in Brazil trace a broad arc moving northeast across the center of Mato Grosso, the southeastern corner of Pará, and the western margins of Tocantins and Maranhão. Transitional seasonally dry evergreen forests form a complex mosaical belt along this arc between taller, more humid forests to the north and west and drier woodland savannas the cerrado to the south and east (Ratter et al., 1973; Eiten, 1975; Ratter et al., 1978). Within this belt, dry forests and cerrado interdigitate at fine spatial scales depending on topography, drainage, soil water and nutrient status, and patterns of dry season fire (Coutinho, 1982; Ratter, 1992; Klink et al., 1993).
In this paper we describe physiographic and floristic gradients within seasonally dry evergreen forests in southeast Pará. The study site is a forest management area called Marajoara (7°50' S, 50°16' W) located 34 km northwest of Redenção, 750 km south of Belém (Fig. 1). Approximately 3 m3.ha-1 of big-leaf mahogany (Swietenia macrophylla, Meliaceae) plus 5 7 secondary timber species were logged from the site between 1992 - 1994. Even though topographic relief within the study area is slight, typically rising only 5 - 8 meters across slopes 200 500 m broad, mahogany's spatial distribution pattern at Marajoara and across the region demonstrates strong positive correlation with seasonal streambeds and low flat terrain adjacent to them (Grogan, 2001; 2003a; 2003b). Other timber species demonstrate similar or inverse distribution patterns (Grogan & Schulze, unpublished data), reflecting community-level structural and compositional differentiation across topographic and edaphic gradients.

Little is known about this region's forests beyond that they sheltered the richest stands of commercial mahogany in Brazil before merchantable stocks were extirpated by the mid 1990s (Grogan et al., 2002). Overland roadway construction in the 1960s and 1970s opened southeast Pará to agricultural settlers, ranchers, gold miners, and loggers specializing in mahogany extraction (Godfrey, 1990; Schmink & Wood, 1992; Veríssimo et al., 1995), situating this region within the so-called Arc of Deforestation which traces the Amazon Basin's southeastern rim. Unlogged forests are rare nowadays on this landscape, most remaining forests have seen second cuts for mahogany and newly merchantable secondary timber species, and forest cover is giving way rapidly to pasture and agriculture at both small-holder and industrial scales. According to regional data published by Brazil's National Institute for Aerospace Research (INPE, 2003), annual deforestation rates during 2000-2001 averaged nearly 6% of the landscape outside Indigenous Areas within the study region (Fig. 1B).
While forest vegetation occurs continuously across topography within the study area, forest structure grades perceptibly across slopes at Marajoara. High-ground forest appears taller, with consistent overstory canopy closure and dense woody vertical structure in the forest understory. Low-ground forest is typically characterized by extremely irregular overstory canopy structure and by denser vines and secondary vegetation in apparently brighter, more frequently disturbed forest understory. (In Fig. 1C, dark areas trace low-ground forests along seasonal streams).
Our objectives are to quantify abiotic and biotic gradients across slopes at Marajoara, describing differences in deep-soil hydrology, soil texture, moisture content, and nutrient status, and forest structure and composition between high and low-ground areas. These observations were prompted by life history studies of mahogany demonstrating non-random spatial distribution strongly associated with low ground adjacent to seasonal streams (Grogan, 2003a). Here we provide environmental context for mahogany's spatial pattern on this landscape a distribution pattern repeated or mirrored by many other commercial timber species at the study site and we suggest reasons why these floristic gradients consistently arise across slopes.
THE STUDY REGION
Climate in southeast Pará is classified as monsoon subtype (Am) of the Köppen system's tropical rain climate (Eidt, 1968) and as tropical dry by Holdridge (1967). Long-term annual precipitation totals range between 1700 1900 mm, increasing towards the north and west. Rainfall distribution is highly seasonal, with a four- to six-month dry season lasting from mid to late May until October or November (Salati et al., 1978).
Gently undulating topographic relief in the study region is shaped by the Brazilian Shield, a Precambrian crystalline bedrock composed of metamorphic and igneous materials gneisses, schists, andesites, granites, basalts which, in their more weather-resistant forms, crop out extensively as isolate or aggregate inselbergs ("island mountains") (Klammer, 1984; Clapperton, 1993). Regional drainage is west-to-east towards the Araguaia River.
Soils in this region are highly diverse. Relatively recent sedimentary formations contact Shield bedrock as well as slowly weathering bedrock extrusions to create a mosaic of red-yellow latosols, eutrophic podzols, and yellow podzols (oxisols, ultisols) (Sombroek & Sampaio, 1962; Radambrasil, 1974). Drainage on low ground may be excessively rapid or impeded, depending on texture and depth to the water table (Sombroek, 1984). Lateritic gravel mixes into soil horizons at unpredictable depths in yellow or red sandy clay soils on slopes (Ab'Saber, 1982).
Transitional dry forests are evergreen with a deciduous component. Radambrasil (1974) classified these as open evergreen forests (floresta ombrófila aberta). Canopies are low and highly irregular, punctuated by scattered emergent mahogany, Apuleia molaris and Hymenaea courbaril (both Caesalpinioideae), and Parkia pendula (Mimosoideae). Brazilnut (Bertholletia excelsa, Lecythidaceae) occurs in this region at extremely low densities. Palms are important components of all forest stories, with the tree-sized babaçu (Attalea speciosa) and inajá (Attalea maripa) occurring at high densities across extensive areas. Patches of vine forest may coalesce into extensive tangled stands. Baider (2000) and Salm (2004) provide floristic descriptions from sites ca. 200 km west of Marajoara.
MATERIAL AND METHODS
Rainfall was measured daily at Marajoara from October 1995 September 2001 in a Tru-Chek direct-reading rain gauge placed in the center of a former log landing 75 m diameter (0.44 ha). Daily temperature maxima and minima were recorded from December 1996 November 1997 with a Taylor Dual Scale temperature gauge mounted on a wall in the camp shelter.
To monitor seasonal changes in water table depths, 10-cm diameter wells were augered to the water table in September 1996, near the end of that year's dry season, at three topographic positions (low ground, midslope, high ground) across two 200 m slopes (henceforth sites 1 and 2), using mud auger buckets and 1.5-m iron extension rods. Elevation differences between high- and low-ground wells measured 5.8 and 5.3 m at sites 1 and 2, respectively. Wells were lined with perforated 10-cm PVC tubing when water-saturated soil could no longer be removed, to 12 m depth at high-ground positions and 6 m depth at low-ground positions; midslope wells were intermediate in depth. Vertical migrations of the water table were documented weekly through five years by measuring the depth to water using a weighted buoy attached to a distance tape. Readings were taken before mid-day.
Physiographic features were mapped at Marajoara within three logging divisions covering 1035 ha. Division roads and forest trails intersecting at 200-m intervals were globally positioned and then mapped on the ground using compass, distance tape, and clinometer. Seasonal streams were mapped by compass and distance tape from within their banks or along trails cut within five meters of their banks. Soils were mapped based on surficial assessments of color, texture, and presence or absence of gravel, lateritic concretions, and or boulder outcrops at 50-m intervals along 54 km of forest trails.
To compare color, texture, pH and nutrient status of high- vs. low-ground soils, four sites were sampled across slopes 200 400 m broad, including sites 1 and 2 where water table wells were installed. At each site, high-ground sampling positions lay five to eight meters above low-ground positions. At each topographic position within the four sites, soils were augered to successive depths (0 10, 10 30, 30 60, and 60 100 cm) at four locations set 20 m apart on transects run in random directions (high ground) or parallel to streambeds (low ground). Respective soil depths were composited proportionally with respect to depth, by position at each site, and then air dried and sifted to remove the > 2 mm component. The Munsell Color Chart was used to determine colors while in the field (wet). Texture was analyzed using the Bouyoucos hydrometer method. Soil pH in water was measured using a calomel electrode. Available cations and available P were measured by double-acid extraction, with solutions analyzed on a Perkin-Elmer 560 atomic absorption spectrometer. Total P and N were determined by digestion prepared with ammonical nitrogen/BD acid and analyzed on a Technicon Auto-Analyzer II.
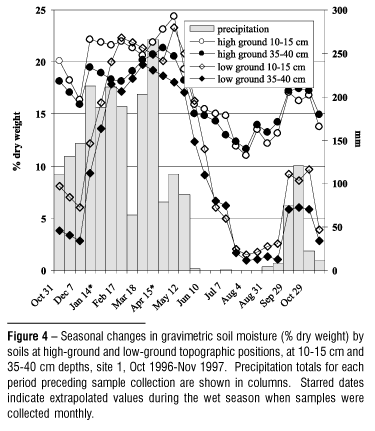
Soil moisture status at high- and low-ground positions at site 1 was monitored gravimetrically as % dry weight from October 1996 November 1997, monthly during the wet season or every two weeks from the late wet season through the dry period. Samples were collected under closed forest canopies where wells to the water table were augered. Soil samples were collected from two depths, 10 15 cm and 35 40 cm. Through the wet season a single sample was collected from each depth at each location; during the dry season two samples were collected from each depth at each location and average % dry weights are reported. Wet and dry weights were recorded to the nearest 0.01 g.
Floristic identifications are derived from a reference collection from Marajoara housed at the IAN Herbarium at EMBRAPA/CPATU, Belém, Pará. Forest composition and structure were sampled at four sites where soils were collected for physical and chemical analyses. At each topographic position, ten 500-m2 circular plots were located on straight-line transects (high ground) or on transects cut in straight-line segments along streams (low ground), turning to follow streams where transects would move >5 m from their banks. Because three of four sampling sites were selectively logged between 1992-1994 for mahogany and secondary timber species, plots were placed at 65-m intervals with fixed rules for re-location where they fell in logged areas. All live trees, palms, and vines > 10 cm diameter (at 1.3 m stem height) were identified (except vines) and measured for diameter. A 100-m2 circular plot was nested around the center of each 500-m2 plot to record live stems and vines 2 10 cm diameter, noting identity (except vines) and diameter. Total area sampled for stems >10 cm diameter was 0.5 ha per topographic position, 1.0 ha per site, or 4.0 ha among four sites split equally between high- and low-ground positions. Total area sampled for stems 2 10 cm diameter was 0.1 ha per topographic position, 0.2 ha per site, or 0.8 ha total, split equally between topographic positions. To describe the rate of canopy gap formation during the 1996-1997 wet season, three surveys were conducted along 16.5 km of trails at Marajoara: in early October before the onset of heavy rains (the baseline survey), in mid February following separate severe windstorms in January and February, and in early June at the beginning of the dry season. During each survey, the linear distance of overhead canopy openings along the trail and the direction of tree or crownfall were measured using Brokaw's (1982) definition for canopy gaps (extending to 2 m above the forest floor). In search of soil charcoal as evidence of forest fires in recent decades or centuries, soil to 50 cm depth was excavated and sifted from six pits 1 m square, split between high- and low-ground positions at sites 1 and 2. Other observations on forest disturbance patterns are anecdotal, based on the authors' residence at Marajoara and discussions with field assistants with many years of experience in this region.
Data analysis
Differences in soil chemical properties and forest structural measures (density, basal area, above-ground biomass) between topographic positions were tested using unpaired t-tests (PROC TTEST, SAS Institute, Cary, NC). Minimum significance was set at a = 0.05. Throughout the text, ± values signify one standard error (SE).
Live biomass was estimated for sampling categories and size classes by the following equations: forest tree species > 10 cm: DW (dry weight) = 0.465*(dbh)2.202 (Overman et al.,1994); forest tree species < 10 cm: log(DW) = -0.85 + 2.57*log(dbh) (Gerwing, 2002); Cecropia sp: ln(DW) = -2.512 + 2.426*ln(dbh) (Nelson et al., 1999); lianas: log (DW) = 0.07 + 2.17*log(dbh) (Gerwing & Farias, 2000). Dbh is stem diameter measured at 1.3 m height.
Importance values (IV) were calculated from inventory data for the aggregated sample incorporating measures of frequency, dominance (through basal area), and distribution (Mori et al., 1983).
The Shannon diversity index H´ was calculated as follows:
H´ = -Epi ln
pi
where pi = proportion of individuals in the ith species
Differences in diversity between topographic positions were tested using Hutcheson's "t" (1970).
A modified version of the Morisita-Horn similarity index (Wolda, 1983) compared forest composition between site positions by asking how quantitatively similar each was to the other. Comparisons were made at three levels: by adjacent site positions, for all combinations of equivalent site positions, and for data composited by position. The formula is:
Cmh = 2 E(ani*bni) / (da + db)(aN*bN)
where ani = the number of individuals in the ith species at position a
aN = the total number of individuals at position a, and
da = Eani2 / aN2
RESULTS
Climate, hydrology, and soils
Mean recorded annual precipitation was 1859 mm during 1995-2001 at Marajoara, with more than 90% falling during seven months from October April (Fig. 2). Annual wet season totals, measured from July June, ranged from 1636 2170 mm. The timing and intensity of wet season onset varied widely from year to year, ranging from early September to as late as mid November. During most years a pronounced lull in rainfall lasting three to four weeks occurred between January and March. Mean monthly temperature maxima ranged from 29 35°C while minima ranged from 17 22°C (Fig. 2).
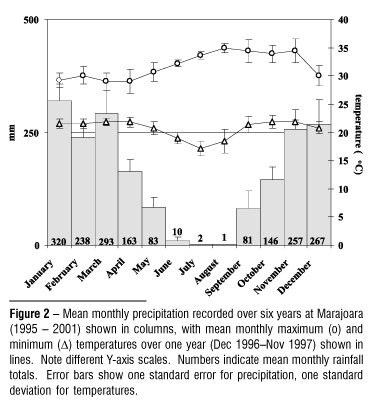
Water table depths across two slopes demonstrated cyclical vertical migrations repeating annually through five years, varying within years according to the seasonal timing, intensity, and quantity of precipitation (Fig. 3). Peak subsidence during the late dry season ranged from 9 12 m below the soil surface at high-ground topographic positions, to 5 7 m depth at low-ground positions, re-establishing each year an essentially horizontal water table by mid to late dry season. Response to early wet season rains or mid wet season dry periods was consistent within and between the two slope series. As groundwater re-charged through the wet season, the water table rose to the soil surface at low-ground positions, persisting there for weeks during the heavy wet seasons of 1996-1997 and 2000-2001. At site 2 the water table at the high-ground position rose nearly to the soil surface during those years.
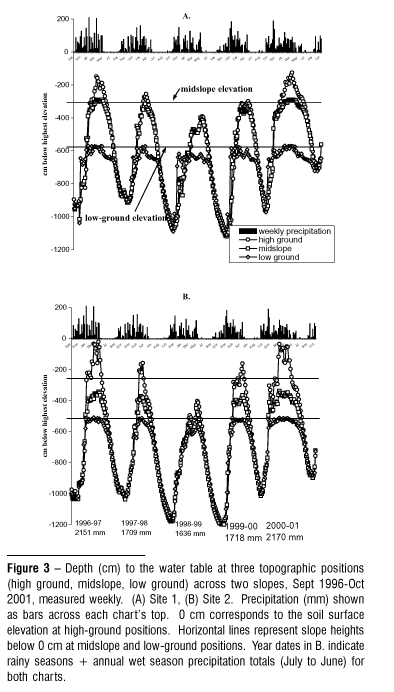
Declines in water table depths began soon after the last significant rains in April or May. Subsidence patterns during the early dry season demonstrate the effect of lateral subsurface flow across topography: water table depths at low-ground positions receded more slowly than at midslope and high-ground positions, remaining within 2 m of the soil surface into the dry season until July or August depending on the previous season's rainfall totals and temporal pattern. After midslope and high-ground water table depths drew approximately even on the horizontal plane with low-ground depths, the water table receded ~30 cm.wk-1 until the wet season's return.
The distribution of soils in 1035 ha at Marajoara illustrates the principal dichotomy on this landscape, with brown, gray, or white sand or sandy loam hydromorphic soils on low ground flanking streambeds and darker brown to red-yellow sand, sandy loam, or clay loam dystrophic soils on high ground distant from streambeds. Based on point sampling along transects, hydromorphic sandy soils covered an estimated 79% of the mapped area. Dystrophic sand or sandy loam soils were common on high ground where slopes rose more than five meters above streambeds. Where slopes rose only 3 4 m between first-order streams, surface soils at slope tops often did not demonstrate textural or color change indicating dystrophic status. Surficial lateritic gravel and lateritic or ironstone concretions (canga) were almost always associated with midslopes.
Textural and color differences between the two principal soil types at Marajoara were consistent at the four sampling sites (Table 1). High-ground soils were sandy at the surface, but clay content increased with depth, yielding sandy clay loam or sandy clay at depth. Soil colors were yellowish brown in surface horizons shifting to strong browns, reddish yellows, and reds below; all chromas were > 4, indicating oxidized soil conditions. These soils are classified as red-yellow latosols or oxisols. Low-ground positions adjacent to seasonal streambeds maintained high sand content to 100 cm depth, with higher silt content in surface (0 10 cm, 10 30 cm) horizons than at depth (30 100 cm). Horizons were gray, grayish brown, or brown above pale, often extremely coarse soils below 30 cm, with chromas > 3 indicating reduced, seasonally waterlogged, hydromorphic conditions. These soils are classified as eutrophic podzols, spodosols, or entisols.
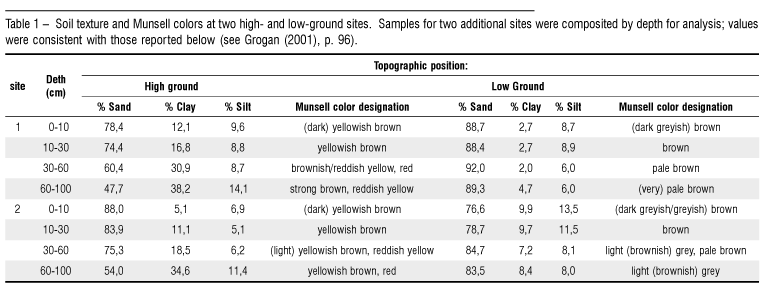
Soil gravimetric moisture content differed little between topographic positions during the wet season, ranging from 17 24% (Fig. 4). However, low-ground soil dried precipitously during the dry season compared to high-ground soil, declining to as low as 1 3% dry weight compared to 11 12% at the high-ground position. Oscillations in moisture content within seasons reflected response to rains followed by dry periods.
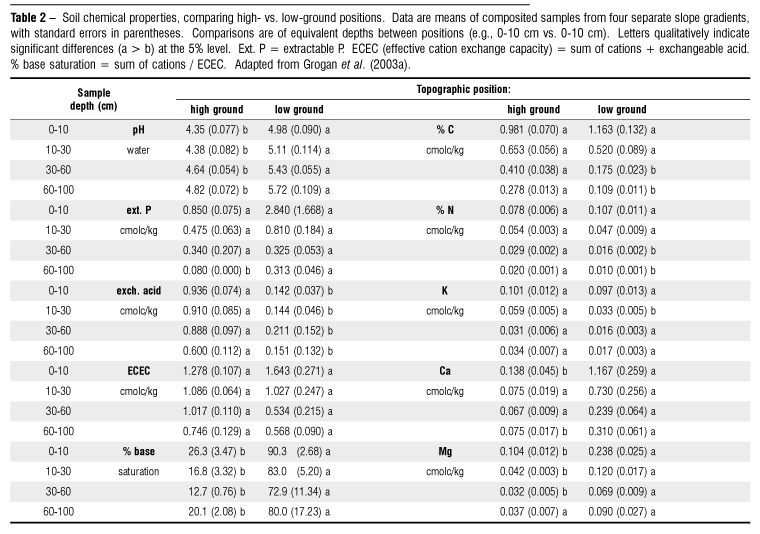
High-ground soils were significantly more acidic than low-ground soils from the surface to 100 cm; soils became less acidic at depth at both positions (Table 2). While shallow horizons at low-ground positions (0 30 cm) had higher mean % C and % N, high-ground soils had significantly higher levels in deeper horizons (30 100 cm). Ca++ and Mg++ concentrations were significantly higher at most depths in low-ground soils, especially surface horizons, with microsite variability at low-ground sites rendering some comparisons non-significant at a = 0.05. Levels of % base saturation were significantly higher throughout low-ground profiles, while the reverse was true for exchangeable acids.
Forest structure and composition
In the 4-ha inventory at four paired high- and low-ground sites, mean density of stems >10 cm diameter was lower on high ground than on low ground (559 vs. 624 ha-1; note non-significant difference, Table 3), due primarily to the clumping palm Euterpe oleracea that typically grows within 5 m of seasonal stream banks. Higher total stem density (> 2 cm diameter) on high ground was due to denser 2 10 cm diameter stems. Mean basal area values for stems > 10 cm diameter were nearly identical between topographic positions (21.5 vs . 21.6 m2.ha-1 ), as were mean live biomass values (256.1 vs. 255.4 Mg.ha-1). Trees > 30 cm diameter were, on average, more frequent on low ground but of smaller stature (diameter). Small vines (< 10 cm diameter) occurred at higher densities on high ground, while large vines (> 10 cm diameter) were more frequent and represented significantly more basal area and live biomass on low ground. Total live biomass for stems > 2 cm diameter was estimated as 290.2 ± 42.0 Mg.ha-1 on high ground vs. 281.9 ± 44.8 Mg.ha-1 on low ground.
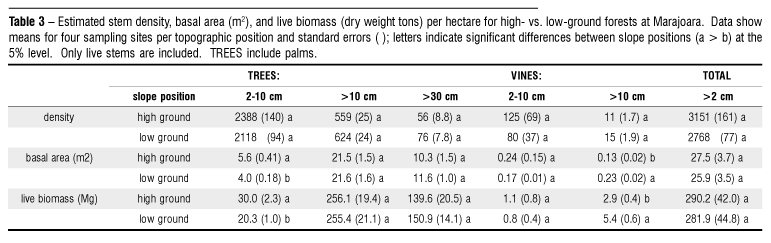
Because three of four sites were selectively logged from 1992-1994, these results may under-represent logged species and underestimate total basal area and live biomass per hectare. Examining data for stems > 10 cm diameter at the unlogged site, basal area of low-ground forest was 27% higher than the mean value for the three other sites (26.0 m2.ha-1 vs. 20.4 m2.ha-1). This level of difference was not observed on high ground (22.5 m2.ha-1 unlogged site vs. 21.4 m2.ha-1 mean for three logged sites).
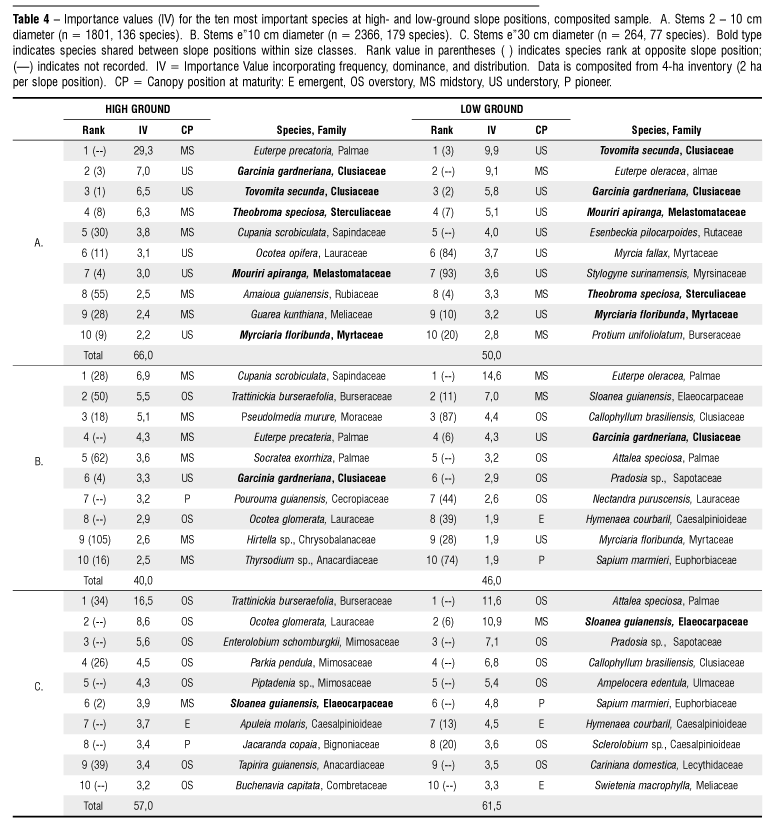
A total of 179 species > 10 cm diameter were recorded in the 4-ha inventory; in 2 ha at each topographic position, 110 species were recorded on high ground vs. 137 species on low ground. The mean number of species per hectare by site was 103 ± 2.2. Among the 136 species recorded in nested plots (2 10 cm diameter, in 0.8 ha total), 36 were not recorded in the >10 cm diameter sample, yielding a total of 215 species encountered during the inventory.
Species importance values (IV) calculated from aggregated data indicate increasing differentiation between topographic positions by increasing stature at plant maturity. Five of 10 most important species in the 2 10 cm sample were shared between high and low ground, four of these shade tolerant small-statured understory trees or treelets (Table 4A). The palms Euterpe precatoria and E. oleracea were ranked 1st and 2nd at high- and low-ground positions, respectively, the former occurring at extremely high densities as single or double stems. In the > 10 cm diameter inventory, only one of the 10 most important species was shared between topographic positions, the shade tolerant site generalist Garcinia gardneriana (Clusiaceae) that was also prominent among smaller stems (Table 4B). That is, "important" species at high-ground positions were markedly less so on low ground and vice versa. This tendency was stronger still for stems > 30 cm diameter, with only one species the shade tolerant Sloanea guianensis (Eleaocarpaceae) ranking in the top 10 at both topographic positions (Table 4C).
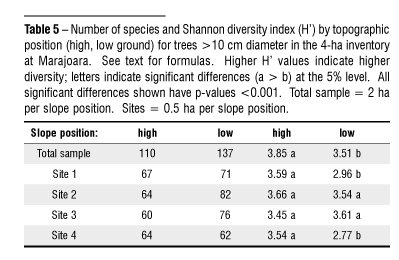
The Shannon diversity index (H´), from which values between 1.5 3.5 are usually calculated, rarely yields values greater than 4.5 (Magurran, 1988). For stems > 10 cm diameter, higher H´ values were assigned to high-ground forests at three of four sampling sites, as well as to the aggregated sample, in spite of generally higher species richness (total number of species) on low ground (Table 5). This is because the Shannon formula emphasizes evenness of distribution as well as species richness: as individuals are concentrated into fewer species, the index returns lower values. The clumping palm Euterpe oleracea, which occurred at high densities on low ground, was largely responsible for these results. According to t-tests, high-ground forests were significantly more diverse (p > 0.001) at two of four site pairs and for the aggregated sample.
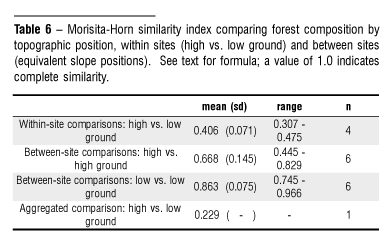
The Morisita-Horn similarity index returns a value between 0 1.0, with higher values representing greater similarity. With one exception, comparisons of equivalent topographic positions between sampling sites indicated greater similarity than high-ground/low-ground comparisons within sites (Table 6). Paired low-ground comparisons yielded higher similarity values, on average, than paired high-ground comparisons. The aggregated sample comparing data pooled by topographic position for all sites yielded the lowest similarity value.
Forest disturbance
Transect surveys of canopy gaps formed during the heavier than average 1996-1997 wet season documented 87 separate canopy disturbances affecting 434 m of trails or 2.6% of transect totals. The largest event covered 30 m. Two convectional storms at mid-season (in January and February) accounted directly for 72% of these events and more than 90% of the transect distance affected. Nearly half (46%) of all affected trees and crowns fell between 241 300°, that is, westwards the two windstorms, as most weather on this landscape does, blew in from the east. Both storms felled many emergent trees, especially Hymenaea courbaril (Caesalpinioideae) and Trattinickia burseraefolia (Burseraceae), the majority of these trees on midslopes or high ground. Most events were single treefalls with collateral damage, that is, smaller trees fallen beneath single large ones. An additional wave of tree- and branchfalls occurred during heavy late wet season rains with little or no winds when soils and tree crowns were water saturated, reducing stability. Vine-laden crowns were especially prone to tip-up or disintegration during this period.
Charcoal was present at various depths in each of six soil pits dug to 50 cm, most of it in bits smaller than 2 mm length, indicating that these forests may burn during excessively dry years, or following successive abnormally dry years. No indirect evidence of Amerindian presence within the study area pottery shards, tools, etc. was encountered during these or any other soil-related research activities at Marajoara.
DISCUSSION
Hydrology and soils
Askew et al. (1970a; 1970b; 1971) and Ratter et al. (1973) described community-level changes in vegetation across soil catenas in the Xavantina-Cachimbo region of northeast Mato Grosso, located approximately 800 km southwest of the study area in similar but drier ecosystems. These authors attributed soil differentiation across gentle slopes in large measure to seasonal water table fluctuations. They concluded that species composition is a good guide to soil nutrient status, with evergreen communities differentiating clearly between hydromorphic soils on low ground and dystrophic or nutrient-poor soils on slopes above them.
Seasonal vertical migrations of the water table at Marajoara are more extreme than those described at South American woodland savanna sites (Rawitscher, 1948; Eden, 1964; Askew et al., 1971; Foldats & Rutkis, 1975; Sarmiento & Vera, 1977). Dense vertically stratified evergreen forest canopies place greater transpirational demand on deep soil water reserves than grass-dominated ecosystems (Poels, 1987; Nepstad et al., 1995; Jipp, 1998); we recovered roots as deep as 12 m while augering water table wells at high-ground positions. Water tables perched at the soil surface for long periods during the wet season on low ground, forcing a shallow, laterally spreading rooting pattern on trees growing there (Lieffers & Rothwell, 1986; Sena Gomes & Kozlowski, 1988); we rarely observed taproots on windthrown trees on low ground. Shallow rooting may in turn expose trees to prolonged soil water deficits during low-rainfall years. The sharp rise in measured water table depths on low ground at the onset of each rainy season at site 1 (Fig. 3A) was caused by lateral flow above indurated pans by heavy early season rainfall.
Successive wet season precipitation totals of 1709 mm and 1636 mm during the 1997-1998 El Niño Southern Oscillation events were not sufficient to recharge water tables to 1996-1997 levels. Highest and mean water table heights fell from one year to the next, suggesting that prolonged drought for example, five to ten years of below-average rainfall could lead to community-wide moisture stress as available deep-soil water recedes below depths to which plants are capable of extracting it to maintain dry season transpiration (Nepstad et al., 1995; 1999). That this trend did not continue during 1999-2000, when wet season rainfall totaled 1718 mm, was likely due to prolonged heavy rainfall from January through March, that is, intra-annual temporal distribution also influences water table recharge.
Soil color and textural changes were consistent with those reported from studies in similar ecosystems on Precambrian bedrock in South America and Africa (Morison et al., 1948; Askew et al., 1970a; 1971; Lawson et al., 1970; Gartlan et al., 1986). The vertical migration and lateral flow of water across slopes are likely the principal factors driving the evolution of soils in this region (Askew et al., 1971). At high-ground positions, argillic (transported clay) horizons below 30 cm may arise both from gravitational transport of fine materials by percolating water and from their export from surface horizons with lateral flow. Indistinct horizonation indicates low organic matter content and intense weathering. Increasing silt and clay content with depth slows infiltration, restricting lateral flow during rainstorms to surface horizons (Young, 1976; Poels, 1987). Rainstorms generate lateral flow across slopes by forcing water through permeable surface horizons that deepen to one m moving downslope, as demonstrated by Poels (1987) in Suriname for rain events exceeding 30 mm, and as observed at Marajoara when streams swell rapidly during wet season storms in the absence of surface flow. At slope bottoms where relief is slight, elevated silt content in surface (< 30 cm depth) horizons indicates illuvial deposition from higher ground (Morison et al., 1948; Young, 1976). A combination of processes could explain coarse sand with low silt content below 30 cm depth to the layer of quartz gravel at ~ 100 cm: removal of fine materials by lateral flow (eluviation), clay decomposition as Fe is leached during alternating oxidative and reductive acid cycles, or podzolization as mobile organic matter forms Al- or Fe-complexes and either precipitates at lower depths or exits with drainage water (Bormann & Likens, 1979; Poels, 1987). Differential removal of fine soil fractions across slopes deepens relief and accelerates the cycle (Poels, 1987).
The texture and nutrient status of low-ground soils may therefore depend on how much lateral flow they are subject to and how long waterlogged conditions persist during the wet season. That is, low-ground soils may differentiate predictably along successively larger streams. Because low-ground soils adjacent to first-order streams draining small areas will be subject to less lateral flow and shorter waterlogged periods than low-ground soils adjacent to higher-order streams, they should be, on average, finer textured and exhibit higher nutrient status because eluvial and leaching processes are weaker. For these reasons we predict that cabeceiras low-ground areas where streams originate will typically have the richest soils on this landscape. Soils there drain better because they are slightly elevated, they remain wet longer into the dry season due to finer texture, and they are nutrient-rich because nutrients transported across slopes with lateral flow tend to accumulate rather than to decompose and flush downstream. Textural differences between low-ground soils at sites 1 and 2 support this hypothesis (Table 1) low-ground sampling positions at site 2 lay 60 m from the start of surface drainage vs. 320 m at site 1. We observed that cabeceiras at Marajoara are often characterized by darker soils (i.e., more nutrient-rich (Grogan, 2001)) and denser, more closed forests compared to soils and communities along higher-order streams.
Smallholder agriculturists with experience farming soils of this region distinguish low-ground soils adjacent to cabeceiras from those adjacent to second- and higher-order streams. The former, they say, offer farmers two advantages: an exceptionally nutrient-rich surface horizon (capa), and higher subsurface clay content that holds more water during the dry season. It may be no coincidence that mateiros exploring unlogged forest for mahogany gravitate toward cabeceiras, knowing from experience that densities of commercial-sized trees are higher there than anywhere else on this landscape.
Forest composition and structure
Species diversity recorded at Marajoara (103 ± 2.2 ha-1 >10 cm diameter) was nearly identical to values reported by Baider (2000) and Salm (2004) from slightly wetter forests 185 km west of the study area. While the two sites shared many of the same species, patterns of dominance were quite different.
Forest inventory data identify the principal constituents of distinctive communities that grade perceptibly one into the other moving across slopes. High-ground communities may be characterized by deeply rooting species performing well in soils that are nutrient-poor compared to those at low-ground positions we observed taproots on some, though not all, windthrown trees on high ground. The deep-rooting habit in well-aerated soils may leave high-ground species less vulnerable to drought.
Low-ground communities may be characterized by species tolerant of alternating swamp- and desert-like conditions as water tables perch and then plummet through wet and dry seasons. Waterlogged wet season soil conditions restrict rooting to sandy surface horizons that hold little water against gravity in the dry season, forcing pursuit of receding water tables by fine roots through dense gleyed subsurface soils. We recorded high rates of drought-related mortality among common low-ground species nearing the end of the El Niño dry season of 1998. Drought conditions extending over several years may cause differential mortality between communities. Though water relations are more extreme, surface horizons of low-ground soils are richer in macronutrients, especially Ca++ and Mg++, providing the boost for early growth that many species, including mahogany, may require for successful recruitment (Grogan et al., 2003a).
Forest disturbance
Continuous pit and mound microtopographic relief common across large flat low-ground areas one to several hectares in size at Marajoara has not been observed on high ground within the study area. This might suggest that windthrow events occurring at large spatial scales are more frequent on low ground, a consequence perhaps of shallow rooting caused by seasonally perched water tables. However, no evidence, such as widespread debris within one or more of these areas, was found during six years of fieldwork at Marajoara to indicate that windthrow is more common or occurs at larger spatial scales on low ground. During the two violent convectional storms of early 1997, trees were windthrown at midslope or high-ground positions at higher than expected rates, either because they stood more exposed to winds there, because fewer possibilities existed for low-ground emergent trees to windthrow after most large mahoganies were removed in 1992-1994, or because of the unpredictable nature of highly local windstorms.
Pit and mound microtopography likely arises from many single-treefall events with collateral damage occurring over many years, and not from rare large-scale blowdowns. Pits on low ground fill with water for prolonged periods during high rainfall years, and at least temporarily during drier years. Few local tree species are capable of colonizing sites where water stands (Grogan, 2001). Because colonization rates are slow, pit and mound relief persists over time, increasing the proportion of surface area in pits which in turn further reduces colonizable area, creating more open canopy conditions that encourage invasive disturbance species like grasses, sedges, and ground herbs, the root-suckering Phenakospermum guianensis (Strelitziaceae), babaçu, woody vines, and late secondary species like mahogany.
Vines play an important role shaping disturbance regimes at Marajoara. During the late wet season the sound of tree and branchfalls is common in closed forest, many of these falling or snapping under the weight of rain-soaked vines. Up to 50% of adult mortality by mahogany is directly (by pulling trees over under heavy loads) or indirectly (through crown suppression) attributable to vines (Grogan, 2001).
Flooding and associated streambank erosion during peak storm events was observed along the highest order stream flowing through the study site, with some alluvial deposition in adjacent flat areas and canopy disturbance from leaning, falling trees. This contributes to canopy irregularity along streams (Fig. 1C).
Finally, fire may play an important role as disturbance agent on this landscape. Cerrado communities are adapted to and at least in part maintained by fire (Coutinho, 1982). Can we assume that transitional forests bordering fire-prone ecosystems also burn, if less frequently? Water table data indicate that compounding low-rainfall years could threaten drought intolerant species. As inter-annual drought episodes lengthen, accelerating tree mortality could build fuel loads for fires. Radiocarbon-dated soil charcoal collected from forest soils throughout Amazonia have led other authors to conclude that successive strata were laid by burns occurring one to many centuries apart since the last glacial maximum (Sanford et al., 1985; Saldarriaga & West, 1986; Bassini & Becker, 1990; Turcq et al., 1997). The presence of charcoal in soils at Marajoara suggests that fire, whether occurring at intervals decades or centuries wide, does indeed play some role in forest dynamics in southeast Pará (Meggers, 1994).
CONCLUSION
Though topographic relief is slight at the study site in southeast Pará, water table levels, soil water availability and nutrient status, and the composition and structure of forest communities vary predictably across slopes and through seasons. Physiographic and floristic gradients in seasonal evergreen forests at Marajoara are largely shaped by water's temporally variable lateral subsurface movement across gentle slopes. Water enters these systems more or less evenly across space, but exits unevenly as surface (in streams) or subsurface flow, channeled by gravity across topography, its lateral movement both part and product of soil textural gradients arising between high and low ground (Young, 1976; Fetter, 1988; Brady, 1990). With fine silt and clay materials, water may also move nutrients, especially Ca++ and Mg++, from high to low ground, where they either accumulate or leach out of the system directly or indirectly through decomposition under waterlogged soil conditions (Bormann & Likens, 1979). Low-ground areas thus could be envisioned as energy and nutrient sinks, where, because of hydrologic cycles, disturbance likely occurs more frequently than at high-ground positions if not necessarily at larger scales. Higher disturbance frequency within low-ground areas, plus soils with higher nutrient status in the surface horizons, may account in large degree for differences in forest communities as tree species sort across slopes according to complex light, moisture, and nutrient requirements.
ACKNOWLEDGEMENTS
Principal funding support for this research was provided by the USDA Forest Service's International Institute of Tropical Forestry. Support was also provided by USAID Brasil, the Charles A. and Anne Morrow Lindbergh Foundation, and the International Tropical Timber Organization's Fellowship Programme. Generous infrastructural support in southeast Pará was provided by the timber export companies Serraria Marajoara Ltda (Semasa) and Madeireira Juary. Our institutional affiliation in Brazil is through the Belém-based non-governmental organization Instituto do Homem e Meio Ambiente da Amazônia (IMAZON). We thank the Brazilian Ministry of Science and Technology (CNPq) for granting permission to conduct fieldwork. We especially thank curator Regina Célia Viana Martins and field botanist Manoel dos Reis Cordeiro at EMBRAPA/CPATU's IAN Herbarium in Belém for generous assistance with the Marajoara reference collection, permanently housed at the Herbarium. We thank Daniel Markewitz at the University of Georgia Athens for advice on soil sampling and nutrient analyses; Manoel Aviz Nascimento and Daniel Nepstad at the Woods Hole Research Center/Instituto de Pesquisa Ambiental da Amazônia (IPAM) for deep-soil augering equipment; and Mark Cochrane at Pennsylvania (now South Dakota) State University for providing georeferenced spatial data that served as a basis for mapping physiographic features. Rodney Salomão at IMAZON kindly provided maps locating the study site. The lion's share of field data reported here was collected by Miguel Alves de Jesus, Waldemir Ribeiro da Cruz, Manoel Rodrigues Vitorino, and Maria Nascimento Rodrigues. We thank Mark Ashton, Mark Schulze, Edson Vidal, and two anonymous reviewers for comments improving the manuscript.
LITERATURE CITED
Ab'Saber, A.N. 1982. The palaeoclimate and palaeoecology of Brazilian Amazonia. In: Prance, G.T. (Ed). Biological Diversification in the Tropics. Columbia University Press, New York, NY, USA. p. 41-59.
Askew, G.P.; Moffatt, D.J.; Montgomery, R.F.; Searl, P.L. 1970a. Interrelationships of soils and vegetation in the savanna-forest boundary zone of north eastern Mato Grosso. Geographical Journal, 136: 370-376.
Askew, G.P.; Moffatt, D.J.; Montgomery, R.F.; Searl, P.L. 1970b. Soil landscapes in north eastern Mato Grosso. Geographical Journal, 136: 211-227.
Askew, G.P.; Moffatt, D.J.; Montgomery, R.F.; Searl, P.L. 1971. Soils and soil moisture as factors influencing the distribution of the vegetation of the Serra do Roncador, Mato Grosso. In: Ferri, M.G. (Ed). III Simpósio sobre o Cerrado. Editora de USP, São Paulo, SP, Brazil. p. 150-160.
Baider, C. 2000. Demografia e ecologia de dispersão de frutos de Bertholletia excelsa Humb. & Bonpl. (Lecythidaceae) em castanhais silvestres da Amazônia Oriental. PhD thesis, Universidade de São Paulo, São Paulo, SP, Brasil. 231pp.
Bassini, F.; Becker, P. 1990. Charcoal occurence in soil depends on topography in terra firme forest. Biotropica, 22: 420-422.
Bormann, F.H.; Likens, G.E. 1979. Pattern and Process in a Forest Ecosystem: Disturbance, Development, and the Steady State Based on the Hubbard Brook Ecosystem Study. Springer-Verlag, New York, NY, USA. 253pp.
Brady, N.C. 1990. The Nature and Properties of Soils. Tenth Edition. Macmillan Publishing Company, New York, NY, USA. 621pp.
Brokaw, N.V.L. 1982. Treefalls: frequency, timing, and consequences. In: Leigh, E.G. Jr.; Rand, A.S.; Windsor, D.M. (Eds). The Ecology of a Tropical Forest: Seasonal Rhythms and Long-term Changes. Smithsonian Institution Press, Washington, DC, USA. p. 101-108.
Clapperton, C. 1993. Quaternary Geology and Geomorphology of South America. Elsevier Science Publishers B.V., Amsterdam, Netherlands. 779pp.
Coutinho, L.M. 1982. Ecological effects of fire in Brazilian cerrado. In: Huntley, B.J.; Walker, B.H. (Eds). Ecology of Tropical Savannas. Springer-Verlag, Berlin, West Germany. p. 273-291.
Eden, M.J. 1964. The savanna ecosystem northern Rupununi, British Guiana. McGill University Press, Montreal, Canada. 216pp.
Eidt, R.C. 1968. The climatology of South America. In: Fittkau, E.J.; Illies, J.; Klinge, H.; Schwabe, G.H.; Sioli, H. (Eds). Biogeography and Ecology in South America. Dr. W. Junk N.V. Publishers, The Hague, Belgium. p. 54-81.
Eiten, G. 1975. The vegetation of the Serra do Roncador. Biotropica, 7: 112-135.
Fetter, C.W. 1988. Applied Hydrogeology. Second Edition. Merrill Publishing Company, Columbus, OH, USA. 592pp.
Data Downloaded June August 2003.
Jipp, P.H. 1998. Deep soil moisture storage and transpiration from forests and pastures of seasonally dry Amazonia. Climatic Change, 39: 395-412.
Klammer, G. 1984. The relief of the extra-Andean Amazon Basin. In: Sioli, H. (Ed). The Amazon: Limnology and Landscape Ecology of a Mighty Tropical River. Dr. W.J. Junk Publishers, Boston, MA, USA. p. 47-83.
Klink, C.A.; Moreira, A.G.; Solbrig, O.T. 1993. Ecological impact of agricultural development in the Brazilian cerrados. In: Young, M.D.; Solbrig, O.T. (Eds). The World's Savannas: Economic Driving Forces, Ecological Constraints, and Policy Options for Sustainable Land Use, Man and the Biosphere Series Vol. 12. UNESCO Parthenon Publishing, New York, NY, USA. p. 259-282.
Lawson, G.W.; Armstrong-Mensah, K.O.; Hall, J.B. 1970. A catena in tropical moist semi-deciduous forest near Kade, Ghana. Journal of Ecology, 58: 371-398.
Lieffers, V.J.; Rothwell, R.L. 1986. Effects of water table and substrate temperature on root and top growth of Picea mariana and Larix laricina seedlings. Canadian Journal of Forest Research, 16: 1201-1206.
Magurran, A.E. 1988. Ecological Diversity and its Measurement. Princeton University Press, Princeton, NJ, USA. 179pp.
Meggers, B.J. 1994. Archeological evidence for the impact of mega-El Niño events on Amazonia during the past two millenia. Climatic Change, 28: 321-338.
Mori, S.A.; Boom, B.M.; Carvalho, A.M.; Santos, T.S. 1983. Southern Bahian moist forests. Botanical Review, 49: 155-232.
Morison, C.G.T.; Hoyle, A.C.; Hope-Simpson, J.F. 1948. Tropical soil-vegetation catenas and mosaics: a study in the south-western part of the Anglo-Egyptian Sudan. Journal of Ecology, 36: 1-84.
Nelson, B.W.; Mesquita, R.; Pereira, J.L.G.; Aquino de Souza, S.G.; Batista, G.T.; Couto, L.B. 1999. Allometric regressions for improved estimate of secondary forest biomass in the central Amazon. Forest Ecology and Management, 117: 149-167.
Nepstad, D.C.; Carvalho, C.R.; Davidson, E.A.; Jipp, P.H.; Lefebvre, P.A.; Negreiros, G.H.; Silva, E.D.; Stone, T.A.; Trumbore, S.E.; Vieira, S. 1995. The role of deep roots in the hydrological and carbon cycles of Amazonian forests and pastures. Nature, 372: 666-669.
Foldats, E.; Rutkis, E. 1975. Ecological studies of chaparro (Curatella americana L.) and manteco (Byrsonima crassifolia H.B.K.) in Venezuela. Journal of Biogeography, 2: 159-178.
Gartlan, J.S.; Newbery, D.M.; Thomas, D.W.; Waterman, P.G. 1986. The influence of topography and soil phosphorous on the vegetation of Korup Forest Reserve, Cameroun. Vegetatio, 65: 131-148.
Gerwing, J.J. 2002. Degradation of forests through logging and fire in the eastern Brazilian Amazon. Forest Ecology and Management, 157 :131-141.
Gerwing, J.J.; Farias, D.L. 2000. Integrating liana abundance and forest structure into an estimate of total above-ground biomass for an eastern Amazon forest. Journal of Tropical Ecology, 16 :327-335.
Godfrey, B.J. 1990. Boom towns of the Amazon. The Geographical Review, 80: 103-117.
Grogan, J.E. 2001. Bigleaf mahogany (Swietenia macrophylla King) in southeast Pará, Brazil: a life history study with management guidelines for sustained production from natural forests. PhD thesis, Yale University, New Haven, CT, USA. 422pp.
Grogan, J.; Barreto, P.; Veríssimo, A. 2002. Mogno na Amazônia Brasileira: Ecologia e Perspectivas de Manejo. IMAZON, Belém, Pará, Brazil. 64p. (http://www.imazon.org.br).
Grogan, J.; Ashton, M.S.; Galvão, J. 2003a. Big-leaf mahogany (Swietenia macrophylla) seedling survival and growth across a topographic gradient in southeast Pará, Brazil. Forest Ecology and Management, 186: 311-326.
Grogan, J.; Galvão, J.; Simões, L.; Veríssimo, A. 2003b. Regeneration of big-leaf mahogany in closed and logged forests of southeastern Pará, Brazil. In: Lugo, A.; Figueroa Colón. J.C.; Alayón, M. (Eds). Big-Leaf Mahogany: Genetics, Ecology, and Management. Springer-Verlag, New York, NY, USA. p. 193-208.
Holdridge, L.R. 1967. Life Zone Ecology. Tropical Science Center, San José, Costa Rica. 206p.
Hutcheson, K. 1970. A test for comparing diversities based on the Shannon formula. Journal of Theoretical Biology, 29: 151-154.
INPE, 2003. Mapa de desflorestamento da Amazônia Legal (2001), Projeto
Prodes Digital 2000, 2001 e 2002 - Monitoramento da Floresta. Instituto Nacional
de Pesquisas Espaciais, Manaus, AM, Brazil. (www.obt.inpe.br/prodesdigital/prodesd.html).
Nepstad, D.C.; Veríssimo, A.; Alencart, A.; Nobre, C.; Lima, E.; Lefebvre,
P.; Schlesinger, P.; Potter, C.; Moutinho, P.; Mendoza, E.; Cochrane, M.; Brooks,
V. 1999. Large-scale impoverishment of Amazonian forests by logging and fire.
Nature, 398: 505-508.
Overman, J.P.M.; White, H.J.L.; Saldarriaga, J.G. 1994. Evaluation of regression models for above-ground biomass determination in Amazon rainforest. Journal of Tropical Ecology, 10: 207-218.
Poels, R.L.H. 1987. Soils, Water and Nutrients in a Forest Ecosystem in Suriname. Agricultural University, Wageningen, Netherlands. 253pp.
Radambrasil. 1974. Projeto Radam, Programa de Integração Nacional, Levantamento de Recursos Naturais: Geologia, Geomorfologia, Solos, Vegetação e Uso Potencial da Terra. Ministério das Minas e Energia, Departamento Nacional de Produção Mineral, Rio de Janeiro, RJ, Brasil. Folha SB 22.
Ratter, J.A. 1992. Transitions between cerrado and forest vegetation in Brazil. In: Furley, P.A.; Proctor, J.; Ratter, J.A. (Eds). Nature and Dynamics of Forest-Savanna Boundaries. Chapman & Hall, London, UK. p. 417-429.
Ratter, J.A.; Richards, P.W.; Argent, G.; Gifford, D.R. 1973. Observations on the vegetation of northeastern Mato Grosso. I. The woody vegetation types of the Yavantina-Cachimbo Expedition area. Philosophical Transactions of the Royal Society of London. B. Biological Sciences, 266: 449-492.
Ratter, J.A.; Askew, G.P.; Montgomery, R.F.; Gifford, D.R. 1978. Observations on the vegetation of northeastern Mato Grosso. 2. Forest and soils of the Rio Suia-Missu area. Proceedings of the Royal Society of London, B, 203: 191-208.
Rawitscher, F. 1948. The water economy of the vegetation of the "campos cerrados" in southern Brazil. Journal of Ecology, 36: 238-268.
Salati, E.; Marques, J.; Molion, L.C. 1978. Origem e distribuição das chuvas na Amazônia. Interciência, 3: 200-206.
Saldarriaga, J.G.; West, D.C. 1986. Holocene fires in the northern Amazon Basin. Quaternary Research, 26: 358-366.
Salm, R. 2004. Tree species diversity in a seasonally-dry forest: the case of the Pinkaití site, in the Kayapó Indigenous Area, southeastern limits of the Amazon. Acta Amazonica, 34: 435-443.
Sanford, R.L.; Saldarriaga, J.; Clark, K.E.; Uhl, C.; Herrera, R. 1985. Amazon rain-forest fires. Science, 227: 53-55.
Sarmiento, G.; Vera, M. 1977. La marcha anual del agua en el suelo en sabanas y bosques tropicales de los Llanos de Venezuela. Agronomia Tropicale, 27: 629-649.
Schmink, M.; Wood, C. 1992. Contested Frontiers in Amazonia. Columbia University Press, New York, NY, USA. 385pp.
Sena Gomes, A.R.; Kozlowski, T.T. 1988. Physiological and growth response to flooding of seedlings of Hevea brasiliensis. Biotropica, 20: 286-293.
Sombroek, W.G. 1984. Soils of the Amazon region. In: Sioli, H. (Ed). The Amazon: Limnology and Landscape Ecology of a Mighty Tropical River. Dr. W.J. Junk Publishers, Boston, MA, USA. p. 521-535.
Sombroek, W.G.; Sampaio, J.B. 1962. Reconnaissance soil survey of the Araguaia mahogany area. FAO, Commisão de Solos, Belém, Pará, Brasil. 61pp.
Turcq, B.; Pressinotti, M.M.N.; Martin, L. 1997. Paleohydrology and paleoclimate of the past 33,000 years at the Tamandua River, Central Brazil. Quaternary Research, 47: 284-294.
Veríssimo, A.; Barreto, P.; Tarifa, R.; Uhl, C. 1995. Extraction of a high-value natural resource in Amazonia: the case of mahogany. Forest Ecology and Management, 72: 39-60.
Wolda, H. 1983. Diversity, diversity indices and tropical cockroaches. Oecologia, 58: 290-298.
Young, A. 1976. Tropical Soils and Soil Survey. Cambridge University Press, Cambridge, UK. 468pp.
Recebido em 05/08/2006
Aceito em 23/11/2006